Ondansetron HCL
33000 INR/Kilograms
Product Details:
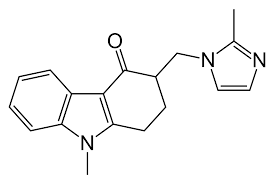
- Molecular Weight 293.4 g/mol Grams (g)
- Molecular Formula C18H19N3O
- Medicine Name Zofran
- Chemical Name Zofran, Ondisolv, others
- CAS No 99614-02-5
- Type Other
- Grade Medicine Grade
- Click to View more
X
Ondansetron HCL Price And Quantity
- 33000 INR/Kilograms
- 25 Kilograms
Ondansetron HCL Product Specifications
- Powder
- 293.4 g/mol Grams (g)
- C18H19N3O
- 99614-02-5
- Other
- Zofran
- Powder
- Zofran, Ondisolv, others
- Ondansetron, marketed under the brand name Zofran, is a medication used to prevent nausea and vomiting caused by cancer chemotherapy, radiation therapy, or surgery. It is also effective for treating gastroenteritis. It has no effect on vomiting caused by motion sickness.
- Medicine Grade
Ondansetron HCL Trade Information
- Mumbai
- 3000 Kilograms Per Month
- 4 Week
- South America Central America Asia Africa
- ISO, WHO
Enter Buying Requirement Details